The Fish Oil Paradox: Benefits, Risks, Evidence
The “Brain Insurance” Myth
If you walk into any health food store today, you’ll see aisles dedicated to Omega-3 supplements. They’re sold as ‘brain insurance’ — the idea that a few capsules a day can shield you from Alzheimer’s, or other forms of cognitive decline and dementia.
It’s a multi-billion-dollar industry built on a foundation of some admittedly promising science. But lately, the narrative has started to shift. We’re seeing large-scale meta-analyses that don’t quite back up the hype. “Meta-analyses” are large studies that combine many clinical trials.
A few recent studies suggest we might actually be taking risks that we didn’t “sign up” for.
Today, I want to walk through some of the actual data — looking at four major clinical reviews, and one specific warning — to get a better handle on whether fish oil is actually protecting our brains, or if you’re just wasting our money on (potentially rancid) piscine oil.
This isn’t medical advice — just a look at what the current evidence actually says.
The Core Theory: Why We Care About DHA
To understand why we’re even talking about this, we have to look at what your brain is made of.
About 60% of your gray matter is fat — by dry weight. And a huge portion of that is a specific Omega-3 fatty acid called docosahexaenoic (/duh-KOH-suh hek-suh-NOH-ick/) acid, or “DHA.”
Think of DHA as a key structural and functional component of neuronal membranes.
According to a 2018 update on Omega-3s and Alzheimer’s [Source 3], these fats do two main things.
Firstly, they keep your cell membranes fluid. These membranes have to be flexible to stay functional when it comes to immune response, nerve signaling, and nutrient transport.
And, secondly, DHA fats act as powerful anti-inflammatories.
Early on, Alzheimer’s is often described as ‘low-grade’ inflammation — like a fire smoldering under the surface. But over time, that can escalate into something sustained and destructive that doesn’t fully resolve..
On paper, Omega-3s should help calm that process.
And, in laboratory and animal studies, Omega-3 has been shown to reduce amyloid-beta accumulation — the plaques long considered hallmarks of the disease.
Though, as we’ll explore in a future video, it’s worth noting that the amyloid hypothesis itself has taken some serious hits recently, including high-profile controversies over foundational research. The science isn’t as settled as supplement labels would have you believe.
And, as we’ve learned in medicine, what happens in a petri dish doesn’t always happen in a human being. Indeed, the “low-grade” label is increasingly being considered an oversimplification.
That said, none of this has stopped supplement suppliers from making promises about fish oil — promises that, as we’ll see, the clinical data doesn’t fully back up.
The Reality Check: What the Data Says
This is where the “matter-of-fact” reality hits. I looked at a systematic review of over a dozen clinical trials [Source 1] and a landmark PubMed review [Source 2] to see if fish-oil supplements actually halt the sort of brain inflammation that develops into full-blown dementia.
Here is the honest breakdown.
If you have, or your loved one has, already gotten a diagnosis of Alzheimer’s Disease, then the supplements don’t seem to do much. The data shows no significant reversal of cognitive decline once the disease is established.
However — and this is a big “however” — some researchers have identified what they call a “window of opportunity.” The stronger evidence points to earlier intervention — pre-clinical stages, or mild cognitive impairment — not after diagnosis.
We’re talking about people with Mild Cognitive Impairment (“MCI”) or people who are cognitively healthy but have a genetic risk, like the APOE4 gene — a genetic variant linked to higher Alzheimer’s risk — which we have gotten into in a previous video.
So, if you’re waiting for memory loss to start before you take your brain health seriously, you’ve likely missed the boat for Omega-3s to be effective.
If it’s anything, it’s better thought of as a preventative tool, not a rescue medication.
And even in studies that do show benefits, the effects tend to be modest — not dramatic.
The Metric: “Test, Don’t Guess”
One of the biggest reasons people see mixed results is that they aren’t actually measuring if the supplement is working. A paper in the journal American Journal of Clinical Nutrition [Source 4] argues that we should be focusing on something called the Omega-3 Index.
Most people just take a pill and hope for the best. But everyone absorbs fats differently. The Omega-3 Index is a blood test. It measures how much EPA and DHA are actually built into your red blood cells — which reflects long-term intake.
An index of ~8% or higher is the established cardiovascular target; brain health research is pointing in the same direction, though that threshold isn’t yet definitive.
If you’re taking a low-quality supplement or your body isn’t absorbing it well, you might still be at a 4% or 5% index — which is basically a deficiency zone.
If you aren’t testing your levels, you’re essentially flying blind. You might be taking a dose that’s too low to matter, or wasting money on a supplement your body isn’t even processing.
You can’t improve what you don’t track.
The Potential Problem: Oxidation and AFib
Now, we should address the “red flag” that’s been popping up in recent headlines.
A report from SciTechDaily [Source 5] highlighted a potential risk that many supplement companies don’t want to talk about: Oxidation.
Fish oil can be unstable. There’s concern that poorly stored or low-quality fish oil may oxidize or, to put it more dramatically, the worry is that it could go rancid when exposed to heat, light, or air.
Plainly, if it turned out that you were swallowing oxidized fish oil, then you wouldn’t be reducing inflammation — you’d potentially be introducing oxidative stress into your system.
But there’s a more specific medical concern. Recent data has linked high-dose fish oil supplementation to an increased risk of Atrial Fibrillation, or AFib — which is an irregular heart rhythm that can lead to strokes.
Now, I want to be careful with my language here, because this finding doesn’t quite fit the definition of an “outlier” that some have used to describe it. An outlier is a single anomalous data point. The AFib signal isn’t that — it has shown up across multiple large clinical trials, which actually makes it a replicated finding. That’s more serious, not less. The supplement industry might prefer you think of this as a fringe result. It isn’t.
This risk appears more pronounced at higher doses and in certain populations.
For example, in this case, the data suggests that “megadosing” fish oil — say, taking 4 or 5 grams a day without a doctor’s supervision — might be creating cardiovascular risks that outweigh the potential brain benefits, especially if you aren’t actually deficient to begin with. Which — again — you won’t know until your levels are measured.
And, to be fully transparent, some risk signals have appeared at lower doses too, particularly in prescription-grade formulations.
The honest bottom line is that the dose threshold isn’t perfectly established yet. Which is all the more reason to loop in your doctor before you dramatically increase your intake.
So yes — there’s a real risk conversation to have here, particularly around dose. But risk in context isn’t the same as “don’t bother.” It just means: be deliberate.
Conclusion: The Pragmatic Approach
So where does that leave us?
Omega-3s clearly matter for brain structure — but supplements aren’t a magic solution.
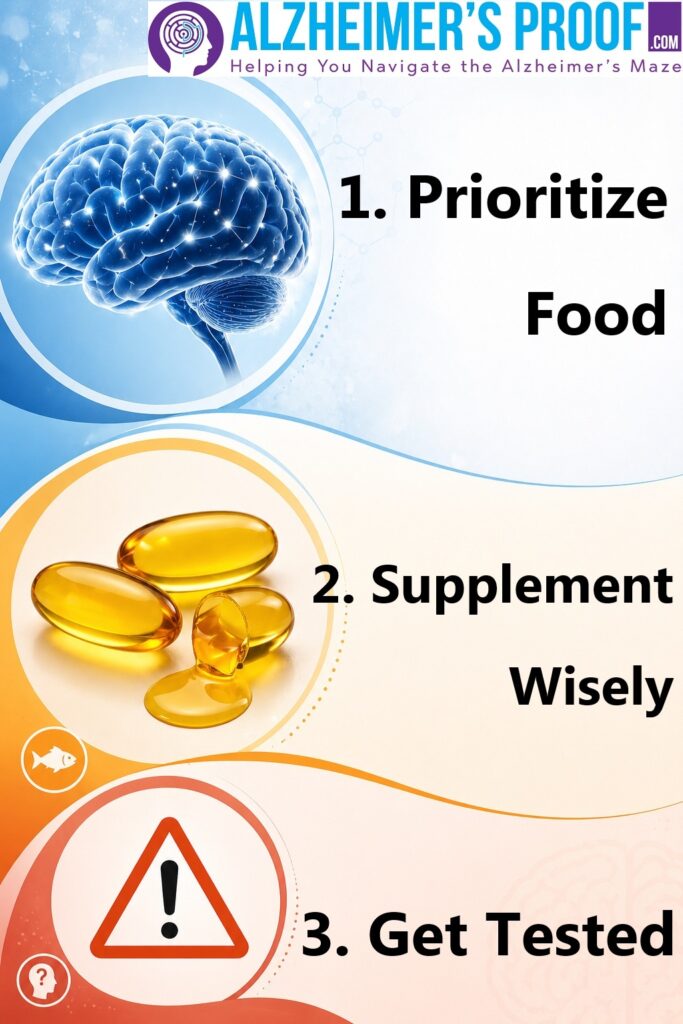
If you’re thinking about using them, a more grounded approach looks like this:
First: prioritize food. Fatty fish like sardines, mackerel, and salmon provide Omega-3s in a more stable, nutrient-rich form.
Second: if you do supplement, be picky. Fish oil is fragile — and quality matters. Choose third-party tested products. Look for labels like the dedicated, so-called “International Fish Oil Standards” program, or “IFOS” (/EYE-fohs/). And… If it smells off, don’t use it.
And third: know your numbers. The Omega-3 Index can tell you whether you’re actually in an meaningful range — or just guessing.
Because in the end, brain health isn’t about one supplement. It’s about maintaining the integrity of your brain over decades.
Fish oil may be one tool — but it’s not the whole toolbox.
If you found this helpful, consider subscribing for future breakdowns like this. And feel free to share it with someone who might benefit.
And, as always, we wish you all the best with your Alzheimer’s-proofing journey. Thanks for watching.
Sources:
[1]: https://pubmed.ncbi.nlm.nih.gov/28986068/ “Effectiveness of omega-3 fatty acid supplementation in patients with Alzheimer disease: A systematic review and meta-analysis – PubMed”
[2]: https://pubmed.ncbi.nlm.nih.gov/19523795/ “Omega-3 fatty acids and dementia – PubMed”
[3]: https://pubmed.ncbi.nlm.nih.gov/30084334/ “A Recent Update on the Effects of Omega-3 Fatty Acids in Alzheimer’s Disease – PubMed”
[4]: https://pmc.ncbi.nlm.nih.gov/articles/PMC9761771/ “The omega-3 index in Alzheimer’s disease: Ready for prime time? – PMC”
[5]: https://scitechdaily.com/scientists-uncover-potential-brain-risks-of-popular-fish-oil-supplements/